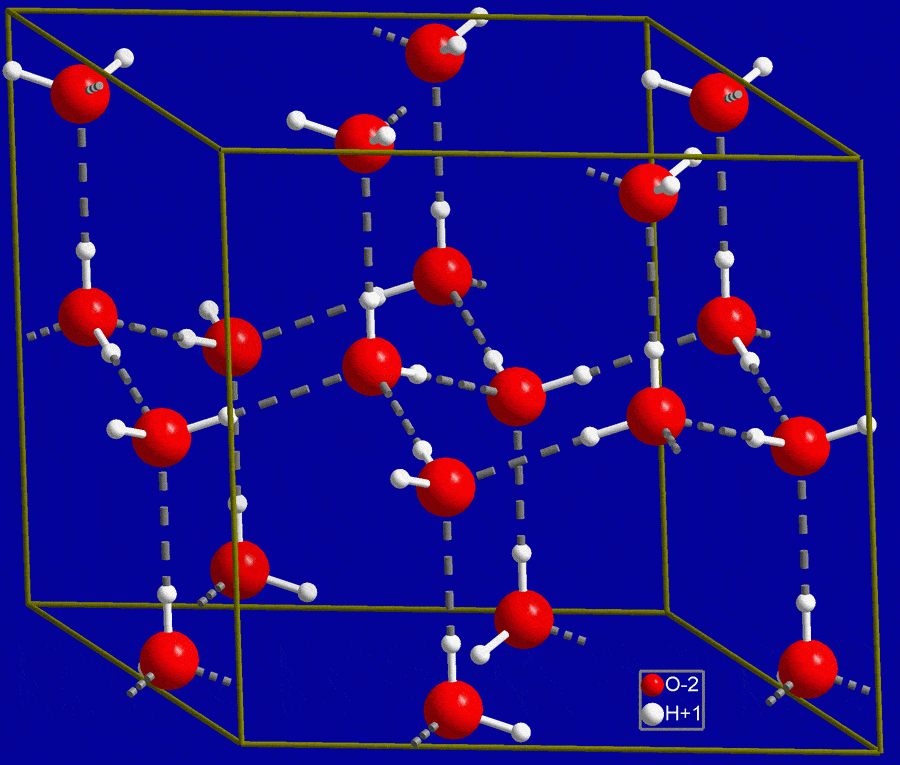
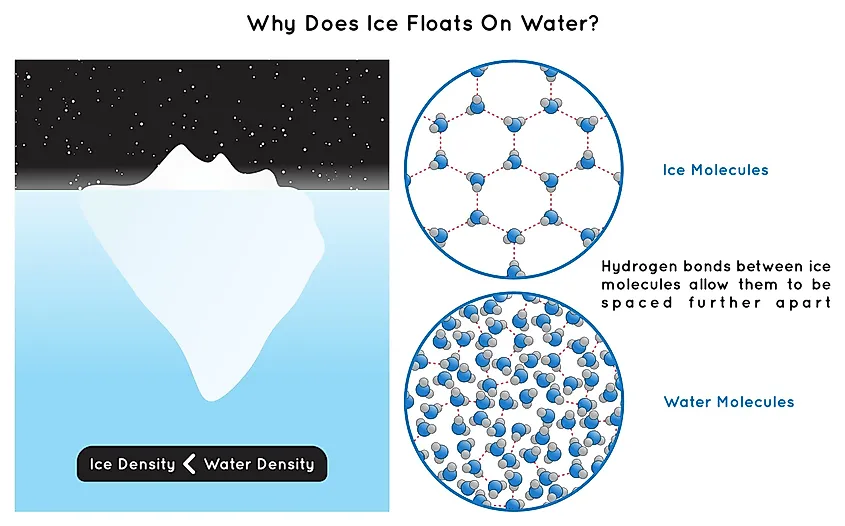
c) N H (d) NH3 14. At its melting point ice is lighter than water because (a) HO molecules are closely packed in solid state (b) Ice crystals have hollow hexagonal

SANOMAT At its melting point ice is lighter than water because 19,0(1)→ No reaction (a) H2O molecules are closely packed in solid state (b) Ice crystals have hollow hexagonal arrangement of H2O

jackeline delrisco on X: "ice is less dense than water,ice molecules are farther apart than water molecules that's why ice floats. https://t.co/9rZoeI8A43" / X

Basic science: understanding experiments: Week 2: 2.2 What if ice was denser than water? | OpenLearn - Open University


:max_bytes(150000):strip_icc()/ice-172152618-5aec5ac1ff1b780036215a57.jpg)